Prof. David Hong provides a summary of the key TRK fusion-positive cancer data presented at ASCO 2022. He selects two abstracts further supporting the profound efficacy and established safety profiles of the first-generation TRK inhibitors (larotrectinib and entrectinib), and a third abstract featuring data for a second-generation TRK inhibitor in a Phase 1 trial with patients resistant to first-generation TRK inhibitors.
NTRK CONNECT Highlights from ASCO 2022
My name is David S. Hong. I am a Professor and Deputy Chair in the Department of Investigational Cancer Therapeutics and Associate Vice President of Clinical Research here at M.D. Anderson Cancer Center in Houston, Texas.
On behalf of NTRK CONNECT, I'm going to present my thoughts on three key abstracts in the previous ASCO 2022 concerning NTRK. The first abstract is by Dr. Alexander Drilon, and that is the long-term efficacy and safety of larotrectinib in a pooled analysis of patients with NTRK fusion cancers. The second abstract is an updated analysis of efficacy and safety of entrectinib in patients with locally advanced, metastatic NTRK fusion positive, solid tumours, and that was by Dr. Maciej Jerzy Krzakowski. And lastly, the third abstract is the safety and pharmacokinetics in clinical efficacy of ICP-723; a highly selective next generation pan-TRK inhibitor in patients with solid tumour from Dr Xiao-Li Wei.
The reason I chose these three abstracts really kind of highlights the activity of these molecules, particularly both larotrectinib and entrectinib. And both with larotrectinib and entrectinib we see that after even several years that the efficacy remains still really profound in patients with NTRK fusion cancers, both with larotrectinib and entrectinib. I would argue though that larotrectinib there seems to be a higher efficacy rate in overall longer follow-up, but both molecules show high activity in patients with NTRK fusion.
This is really highlighted by another abstract that was featured in ASCO 2022 by Dr Bokemeyer that looked at patients who received larotrectinib in kind of a silicon or a computational analysis, with real-world data from a Flatiron database showing that indeed patients with larotrectinib in a match-to-match comparison with similiar patients showed significant overall survival, and a lower risk of death over a similar period of time.
My last poster I want to feature is this abstract by Dr Xiao-Li Wei, looking at really the second-generation of TRK inhibitors. There are now several TRK inhibitors, —second-generation TRK inhibitors in the clinic, in clinical trials, repotrectinib, Pyramid Biosciences also has a TRK inhibitor ongoing. And this TRK inhibitor targets one of the most common mutations that we do see in patients with larotrectinib and entrectinib resistance, the solvent-front mutations. And what Dr Wei showed in these patients with NTRK that indeed there were responses, close to 67% responses in these patients with NTRK fusion.
Thank you again for taking your time to listen to my review. You can download these slides from the website, and again thanks again for listening.
Dr David S. Hong is a Professor, Deputy Chairman in the Department of Investigational Cancer Therapeutics (Phase I Program), Clinical Medical Director of the Clinical Translational Research Center (CTRC), and Associate Vice President of Clinical Research at The University of Texas MD Anderson Cancer Center.
Dr Hong was instrumental in forming one of the largest and most innovative Phase 1 clinical trial units in the world, with over 1300 patients enrolled in clinical trials in FY2021 and over 400 active ongoing clinical trials.
Dr Hong has been the Principal Investigator of over 120 research protocols that involve a wide range of sponsors, including the Cancer Therapy Evaluation Program (CTEP) at the National Cancer Institute. He has published over 374 publications in Peer-reviewed Journals such as NEJM, Lancet Oncology, JCO and Nature.
He has been involved in the early development of cabozantinib, siltuximab, dabrafenib, trametinib, regorafenib, lenvatinib, larotrectinib, tesotumab vendotin, and sotorasib the first drug to target KRAS that have led to FDA approval.
He has also helped found 2 companies OncoResponse and Telperian.
He is an expert on c-Met, NTRK, KRAS, and adoptive cellular therapy in solid tumors and has led several national trials such as the c-Met amplified, c-Met exon 14 deleted, and NTRK arms of the NCI-MATCH trial. He been awarded the ASCO Young Investigator Award, the Jesse Jones award, Best Boss award at MD Anderson, Irwin Krakoff Award for Clinical Research, and the Gerald P. Bodey award for education and Mentor of the year award in the Division of Cancer Medicine, MD Anderson.
AbbVie, Adaptimmune, Adlai-Nortye, Amgen, Astra-Zeneca, Bayer, Bristol- Myers Squibb, Daiichi-Sankyo, Deciphera, Eisai, Endeavor, Erasca, F. Hoffmann- LaRoche, Fate Therapeutics, Genentech, Genmab, Ignyta, Infinity, Kite, Kyowa Kirin, Lilly, LOXO, Merck, Medimmune, Mirati, Mologen, Navier, NCI-CTEP, Novartis, Numab, Pfizer, Pyramid Bio, SeaGen, Takeda, TCR2, Teckro, Turning Point Therapeutics and VM Oncology.
Other programmes of interest
Innovating relapsed refractory multiple myeloma care
Unmet needs, therapy management, and real-world experience
Experts
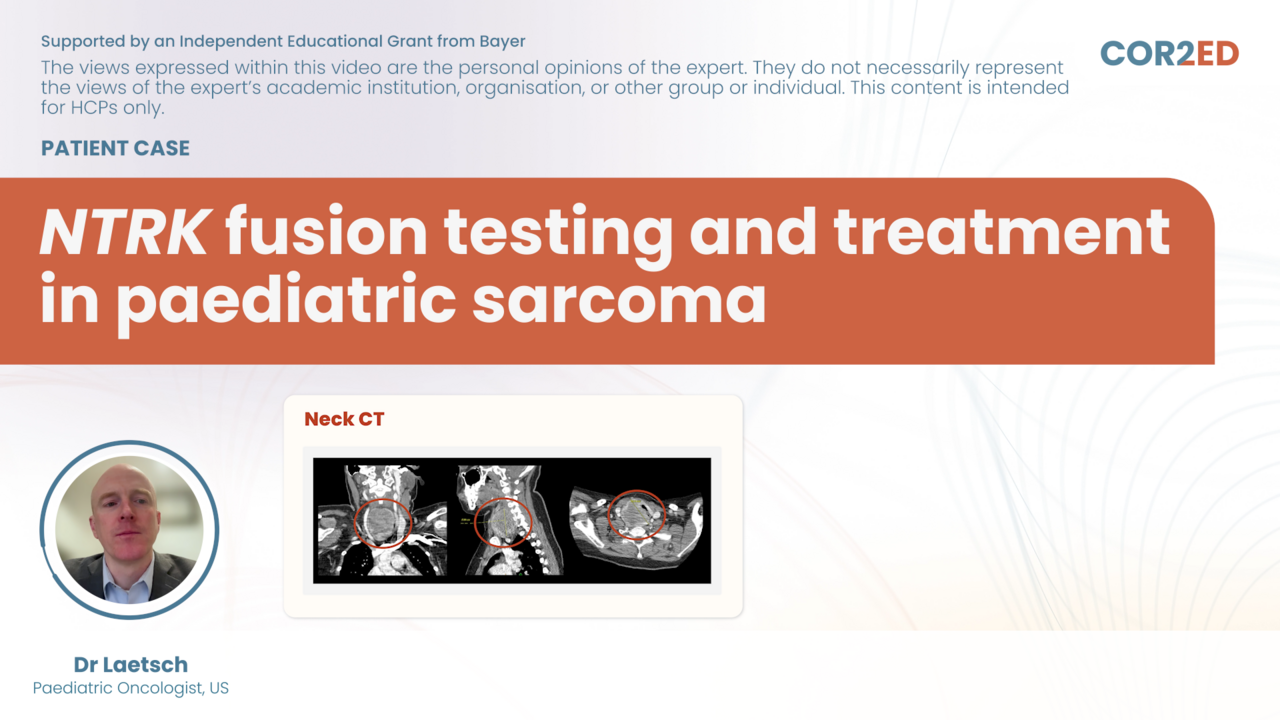
Assoc. Prof. María Victoria Mateos, Assoc. Prof. Karthik Ramasamy, Assoc. Prof. Elena ZamagniNTRK fusion testing and treatment in pediatric sarcoma
Make decisions for a young patient during his cancer journey
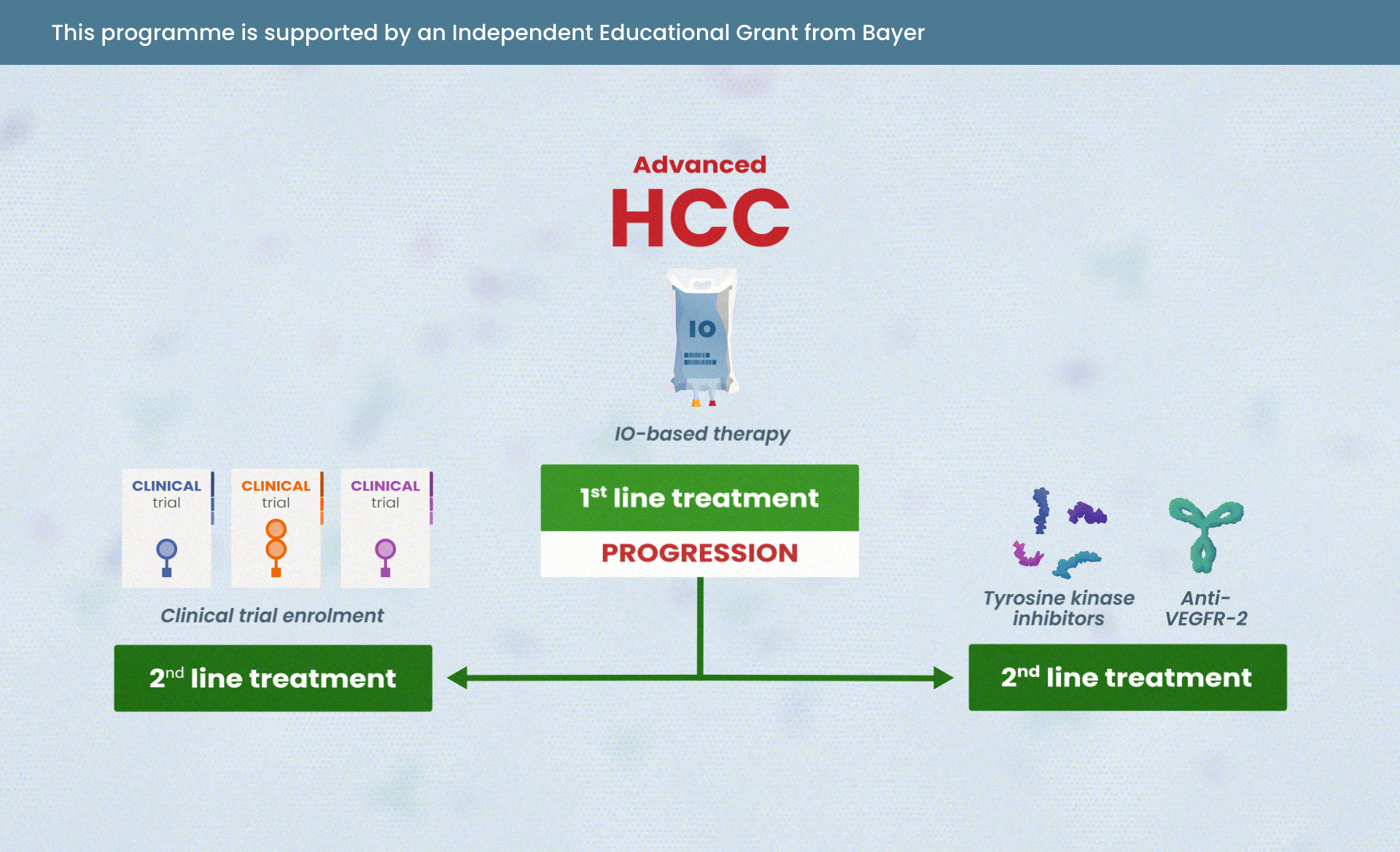
Navigating advanced HCC: treatment strategies beyond immunotherapy (IO)
Module 4: Treatment strategies for patients ineligible for IO or those with progression on IO
Later-line treatment strategies in metastatic colorectal cancer (mCRC)
Rechallenge vs switching





 Downloadable
Downloadable  5 MIN
5 MIN
 Jun 2025
Jun 2025